news - press-releases
New paper published on high multiplexing
Date
We are happy to announce the publication of our latest scientific paper in collaboration with Prof. Dr. Soltermann (University Hospital of Zurich) and Prof. Gijs (Laboratory of Microsystems at EPFL, Lausanne).
The article was published in Microsystems and Nanoengineering, a member journal of Springer Nature, discussing “Microfluidics-assisted multiplexed biomarker detection for in situ mapping of immune cells in tumor sections”.
You can read the full, open-access paper here.
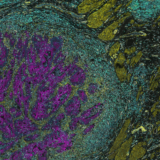
With this study, we provide a proof-of-concept of the use of microfluidics for the automation of high-multiplexed immunostaining. In order to analyze the tumor microenvironment, understanding the spatial organization of multiple biomarkers present on a tissue section is of key importance. With this purpose, it is possible to stain and visualize several targets simultaneously with the technique known as “multiplexed immunofluorescence”, the fluorescent staining of multiple biomarkers on the same tissue sample.
Differently from other single-plex immunostaining techniques, multiplexing allows to save scarce tissue sample material while it enables quantitative analysis such as identifying correlations on several cell families and their activation status within the tumor microenvironment.
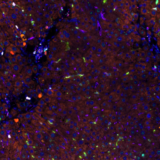
In this study we have demonstrated the potential of our microfluidic staining device to stain 10 markers of relevance in the field of immuno-oncology (CK, CD3, CD4, CD8, FOXP3, CD20, CD68, CD56, PD-1, and PD-L1) in a sequential immunofluorescence protocol consisting of cycles of staining-imaging-elution of antibodies. We have also shown a quantitative approach for the automation of both the experimental and analytical methods with our system.
Lunaphore’s automated device is based on the FFeX technology, utilizing a microfluidic tissue processor capable of staining each marker within 9-11 minutes. This microfluidic chip is equipped with an imaging window. It is therefore referred to as Look-Through-Chip (LTC) and is located in the staining module of the instrument. The imaging system has been integrated in the device and can image through this chip, in order to fully automate the staining-imaging process. The study included as well high-quality image-processing algorithms to map each cell in the entire tissue, identifying the coexpression and colocalization patterns of biomarkers in order to classify the immune cells and their activation status.
“Combining the rapid immunofluorescence staining achieved with our microfluidic technology, together with an integrated microscopy system, we are able to perform high-plex assays using off-the-shelf unlabeled primary antibodies in unprecedented times” said Diego Dupouy, Lunaphore’s CTO. He added: “We are currently developing a high-throughput instrument to run 40-plex assays in a fully automated manner using the method demonstrated in this study”.
Indeed, the advantages of microfluidics in a microscope-integrated multistaining instrument are numerous: 1) It is possible to automate high-multiplexing protocols. 2) The number of markers stained can be very high. In this study we have shown the automation of a 10-plex but the system allows to stain up to 40 markers on one sample. 3) Finally, the use of standard unlabeled primary antibodies will prevent issues related to availability often occurring with the use of labeled antibodies that are required by alternative multistaining techniques.